How Does It Work?
The L360 Thigh System electrically stimulates the nerves of the affected leg’s muscles, providing knee flexion or extension, while also potentially improving the user’s gait.


3D Motion Detection — A proprietary adaptive algorithm adjusts to kinematic changes in real time, deploying stimulation in 0.01 seconds, precisely when needed.
Electrical Stimulation Modes — Targeted stimulation occurs in three distinct modes:
- FES: gait mode exclusive, with proprietary intelligence
- FES: cycle training mode, exclusive with proprietary intelligence
- NMES: training mode

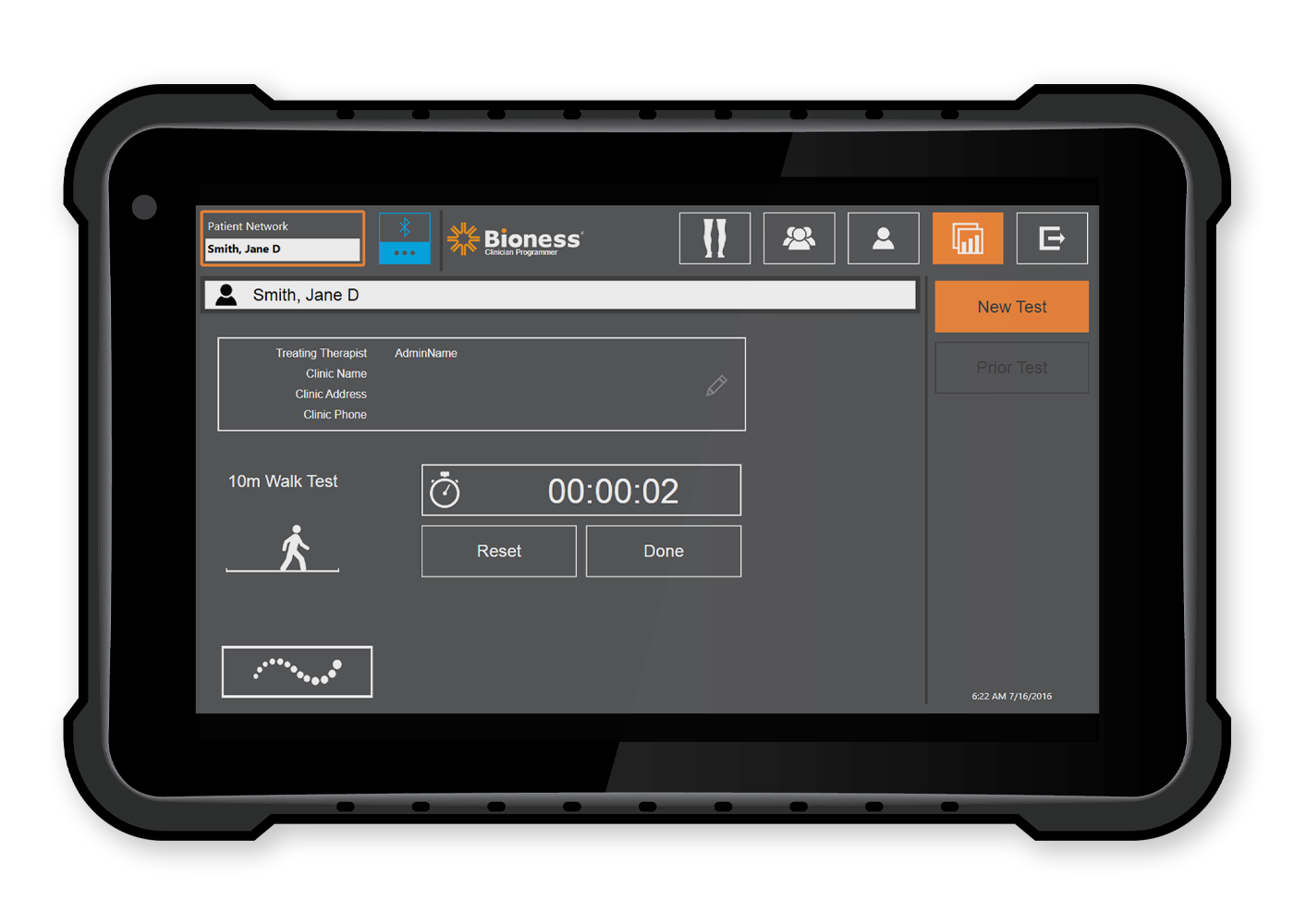
Outcome Measures — Standardized assessments make it easy for clinicians to objectively track and document patient progress.
- 10-meter walk test
- Distance
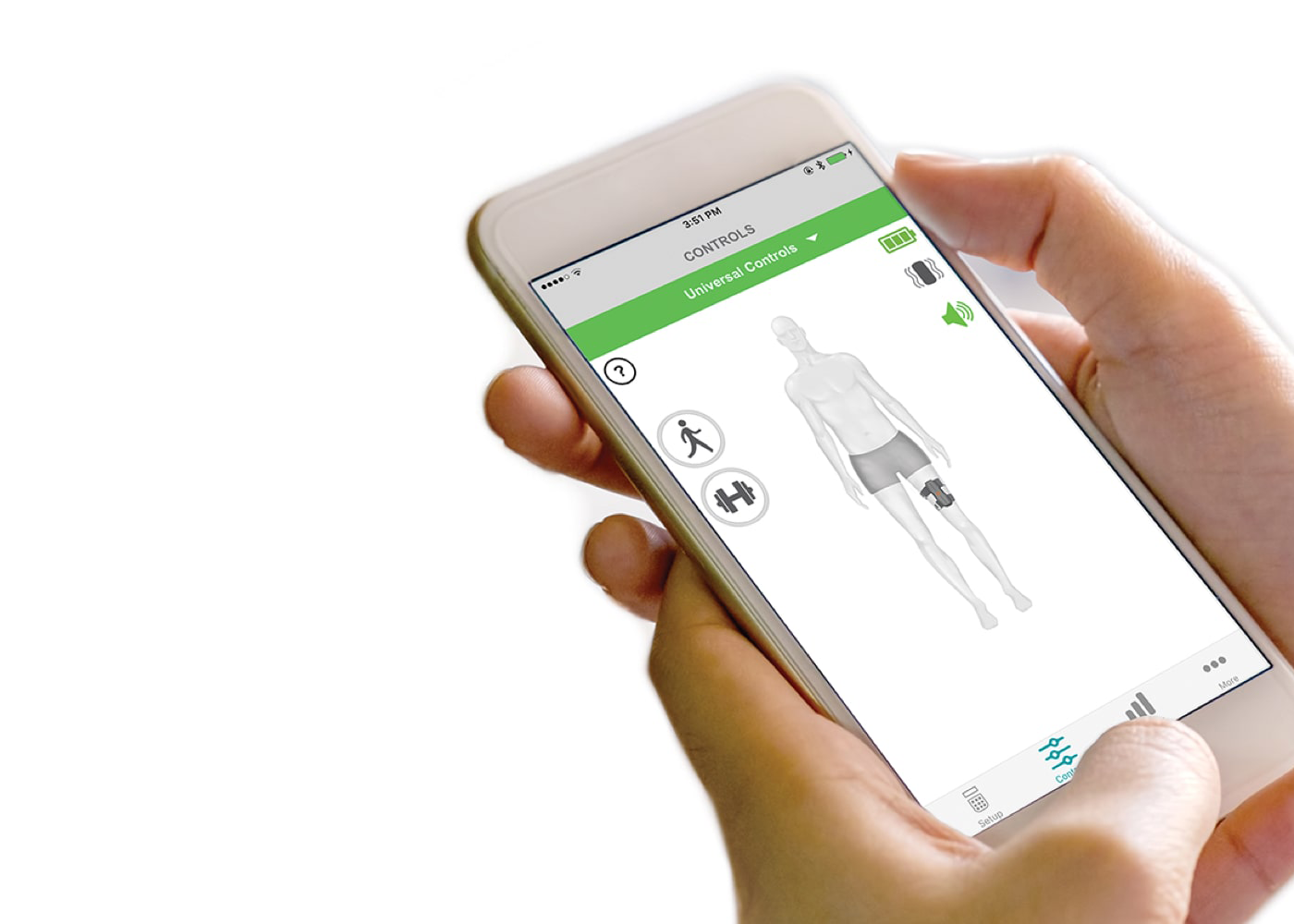
User App – The myBioness™ mobile app lets users control their L360 Thigh System, set goals, and monitor their activity and progress.
Contact us to schedule a demonstration of the L360 Thigh System or to get more information about Bioness products.
Indications for Use: The L360 Thigh System is intended to assist knee flexion or extension in adult individuals with muscle weakness related to upper motor neuron disease/injury (e.g., stroke, damage to pathways to the spinal cord). The L360 Thigh System electrically stimulates muscles in the affected leg to provide knee flexion or extension; thus, it also may improve the individual’s gait.
The L360 Thigh System may also facilitate muscle re-education, prevent/retard disuse atrophy, maintain or increase joint range of motion, increase local blood flow, provide early post-surgical quadriceps and hamstring strengthening, improve post-surgical knee stability secondary to quadriceps and hamstring strengthening, and relax muscle spasms.
L360 Thigh System is contraindicated in patients with a demand-type cardiac pacemaker, defibrillator or any electrical implant. Do not use the system on a leg where metallic implant is directly underneath the electrodes, a cancerous lesion is present or suspected, or on a leg with regional disorder (e.g., fracture or dislocation) which could be adversely affected by motion from the stimulation. Use caution in patients with diagnosed or suspected cardiac problems or epilepsy.
Full prescribing information can be found in product labeling or at: www.bionessrehab.com/l360/safety-information.

