How Does the H200 Wireless Work?
Precise Stimulation, Intuitive Design, Clinical Efficiency

The H200 Wireless Hand Rehabilitation System applies FES in a precise sequence to stimulate the nerves and muscles of the forearm and hand. The stimulation causes the muscles to move and, over time, may help them relearn how to respond when the brain tells the nervous system to move.
Thanks to its wireless design, the H200 Wireless is easy to integrate into clinical therapy sessions and the patient’s daily life, encouraging treatment compliance. For clinicians, that may mean:
- Improved patient outcomes
- Increased patient productivity
- More efficient caseload management
- Shorter in-clinic stays or out-patient treatment programs
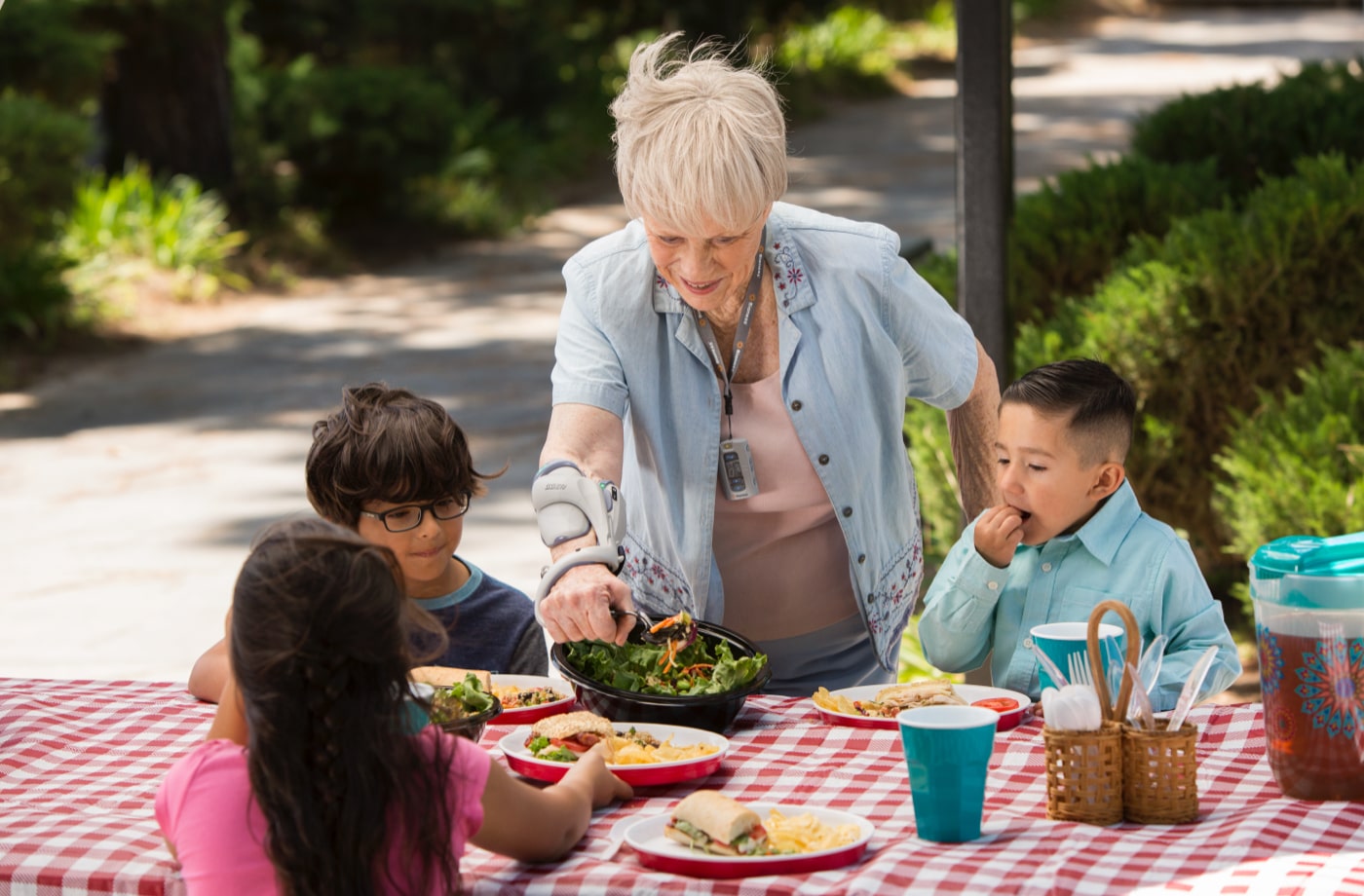
The H200 Wireless system’s components include:

Orthosis

Control Unit
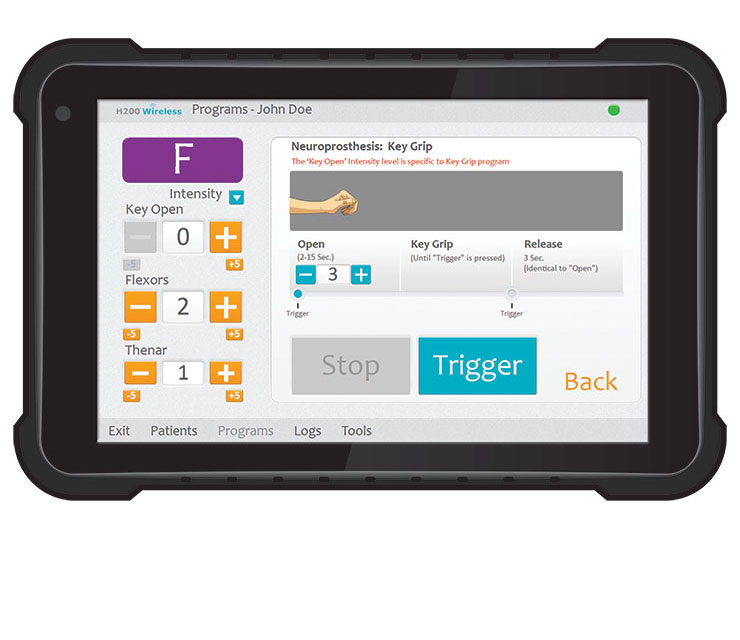
Clinician’s Programmer
A clinician fits the orthosis and sets up the control unit with a patient-specific therapy program. From there, the user can put on the orthosis and use the system as needed to perform activities.
Review the Research Studies
Check out some of the highlights of the clinical evidence about the H200 Wireless Hand Rehabilitation System. Read more on the Reseach Studies page.
Contact us to schedule a demo with H200 Wireless or if you have questions about other Bioness products.
Indications for Use:
The H200 Wireless System is an electrical stimulation device indicated for the following uses:
- Functional Electrical Stimulation (FES)
- Improvement of hand function and active range of motion in patients with hemiplegia due to stroke or upper limb paralysis due to C5 spinal cord injury.
- NeuroMuscular Electrical Stimulation (NMES)
- Maintenance and/or increase of hand range of motion; Prevention and/or retardation of disuse atrophy; Increase in local blood circulation; Reduction of muscle spasm; Re-education of muscles.
H200 Wireless is contraindicated in patients with a demand-type cardiac pacemaker, defibrillator or any electrical implant. Do not use the system on a hand where metallic implant is directly underneath the electrodes, a cancerous lesion is present or suspected, or on a hand with regional disorder (e.g., fracture or dislocation) which could be adversely affected by motion from the stimulation. Use caution in patients with diagnosed or suspected cardiac problems or epilepsy.
Full prescribing information can be found in product labeling or at www.bionessrehab.com/h200/safety-information/.
Instructions for Use / Patient Guides and Reference Cards can be made available upon request.
Contact [email protected] or 800-211-9136 to request an electronic copy.

